Abstract
Direct deoxyazidation of alcohols with NaN3 is a straightforward method for the synthesis of widely used alkyl azides in organic chemistry. However, known methods have some limitations such as high reaction temperatures and narrow substrate scope. Herein, a general and practical method for the preparation of alkyl azides from alcohols using NaN3 has been developed. N-tosyl-4-chlorobenzenesulfonimidoyl fluoride (SulfoxFluor) plays an important role in this deoxyazidation process, which converts a broad range of alcohols into alkyl azides at room temperature. The power of this deoxyazidation protocol has been demonstrated by successful late-stage deoxyazidation of natural products and pharmaceutically relevant molecules.
Similar content being viewed by others
Introduction
Organic azides are a class of important compounds that have been used as precursors for nitrenes and in the synthesis of amines, and more popularly in the copper-catalyzed azide-alkyne cycloadditions (known as click chemistry)1,2,3,4,5,6,7,8,9,10. Alkyl azides are typically prepared by nucleophilic substitution (SN2) with an azide ion (N3−), and the diazo-transfer reaction to primary amines using triflyl azide (CF3SO2N3) or fluorosulfuryl azide (FSO2N3) has emerged as a powerful method for the preparation of alkyl azides from primary amines1,11. On the other hand, given the ready availability of structurally diverse alcohols, direct conversion of alcohols to alkyl azides via deoxyazidation would be an attractive synthetic strategy, which avoids the use of genotoxic alkyl halides and sulfonates in azidation reactions12. Unfortunately, the alcoholic hydroxyl group is a poor leaving group, so its direct displacement by azide ion is generally difficult. Previously, Mitsunobu conditions have been investigated by using different azide ion sources such as HN3, TMSN3, (PhO)2P(O)N3, Zn(N3)2•2Py, or n-Bu4NN3, but the Mitsunobu conditions are not amenable to the most readily available and cost-effective azide source—NaN3 (Fig. 1, Eq 1)13,14,15,16,17,18,19,20,21,22,23,24,25,26. Indeed, the currently known NaN3-based deoxyazidation methods are sparse (Fig. 1, Eq 2). Both NaN3/BF3•Et2O27 and NaN3/triphosgene28 systems are only applicable to allylic and benzylic alcohols, and other NaN3-based methods (using NaN3/TsIm29, (2,4-Cl2C6H3O)2P(O)Cl/NaN330, or halocarbon/Ph3P/NaN331,32,33,34) suffer from the high reaction temperatures and/or narrow substrate scope. Therefore, the development of a general method for efficient and direct conversion of alcohols to alkyl azides with NaN3 is highly desirable.
a Illustration of previous work on deoxyazidation of alcohols (Eqs 1–2) and deoxyfluorination of alcohols with SulfoxFluor (Eq 3). b Illustration of this work. Eq 4 refers to the SulfoxFluor-mediated deoxyazidation of alcohols with NaN3. Eqs 1–3 refer to the previously reported deoxyazidation of alcohols (previous work), and eq 4 shows the SulfoxFluor-mediated deoxyazidation of alcohols with NaN3 (this work).
Sulfonimidoyl compounds possess diverse reactivity (compared with sulfonyl compounds) due to the modulation by the nitrogen substituent35,36,37. During the past decade, our group has been interested in developing fluoroalkyl sulfoximines as versatile fluoroalkylation reagents38,39. Recently, we reported that N-tosyl-4-chlorobenzenesulfonimidoyl fluoride (SulfoxFluor) can serve as a deoxyfluorination reagent for converting alcohols to alkyl fluorides (Fig. 1, Eq 3)40. In this fluorination process, the in situ formed alkyl sulfonimidate (from SulfoxFluor and alcohol) serves as the real electrophilic alkylating agent to react with hydrogen-bonded fluoride ion, affording the desired alkyl fluoride40,41. We envisioned that since fluoride ion is a weak nucleophile42, if there is a strong nucleophile (namely, azide ion) existing in the reaction system, the deoxyazidation of alcohol could become the dominating reaction pathway, giving an alkyl azide as a major product.
Herein, we show a general and practical protocol for deoxyazidation of readily available alcohols with NaN3 using SulfoxFluor as an activator (Fig. 1, Eq 4). A wide range of alkyl azides could be obtained successfully under mild reaction conditions.
Results
Optimization of reaction conditions
At the onset of our investigation, we chose the primary alcohol 2a as a model substrate, NaN3 as an azidation agent, SulfoxFluor as an activator, 1,8-diazabicyclo-[5.4.0]undec-7-ene (DBU) as a base40,43,44,45,46, and DMF as a solvent; and the reaction was carried out at room temperature. The preliminary result showed that the use of SulfoxFluor (1.0 equiv) afforded azide 3a in 59% yield and a majority of 2a (31%) remained (Table 1, entry 1). Further optimization of the equiv of SulfoxFluor, NaN3, and DBU showed that azide 3a was formed in 84% yield without fluorination and elimination by-products (Table 1, entry 4). Reducing the equiv of NaN3 resulted in the formation of alkyl fluoride 4 (Table 1, entries 5–6). No azide 3a was formed when triethylamine and pyridine were used as bases (Table 1, entries 7–8). For secondary alcohol 2b, it was found that the use of 2.2 equiv of SulfoxFluor was not enough, and the desired alkyl azide 3b was formed in only 38% yield, along with a significant amount of 2b (44%) remained (Table 2, entry 1). Changing the amounts of both SulfoxFluor (2.8 equiv) and DBU (4.0 equiv) resulted in a higher yield (65%) of 3b (Table 2, entries 2–4). Further screening of the reaction conditions showed that an 84% yield of 3b could be obtained in 12 h by performing the reaction with 2b (1.0 equiv), NaN3 (2.0 equiv), SulfoxFluor (2.8 equiv), and DBU (4.0 equiv) at room temperature; and remarkably, alcohol 2b was completely consumed and no fluorination and elimination side products were formed (Table 2, entry 5). Notably, the use of perfluorobutanesulfonyl fluoride (instead of SulfoxFluor) resulted in a decrease of the yield of 3b (68%), with 7% of elimination side product 7 being formed (Table 2, entry 6)45,46,47. Shortening the reaction time to 6 h or using other solvents (such as DMSO, toluene, and CH3CN) did not give better yields of product 3b (Table 2, entries 7–10).
Comparison of various sulfonyl fluorides and sulfonimidoyl fluorides in deoxyazidation of alcohols
To demonstrate the uniqueness of our reagent in the deoxyazidation reaction, several sulfonyl fluorides and sulfonimidoyl fluorides were compared to show their reactivity. 2,2,2-Trifluoroethanol (2c) was chosen as a model substrate to react with these reagents under standard conditions. The results are shown in Table 3. An excellent yield of azide 3c (93%) was formed by using SulfoxFluor as an activator, along with a small amount of 2c (4%) remained (Table 3, entry 1). Changing the S-substituent to an electron-neutral or more electron-deficient 4-nitrophenyl group resulted in a decrease in the yield of 3c (Table 3, entries 2 and 3). Moreover, in the case of 1d with an N-alkyl substituent, no azide 3c was formed and nearly half of 2c was converted to the sulfonimidoyl ester intermediate 8d (Table 3, entry 4). When 2-pyridylsulfonyl fluoride (PyFluor) 1e and tosyl fluoride 1f were used, a full conversion to the corresponding sulfonyl ester intermediates was observed (Table 3, entries 5 and 6). Replacing the N-substituent from tosyl to tertiary butyl led to no azide formation, and a recovery of 2c (82%) was observed (Table 3, entry 7). In the case of perfluorobutanesulfonyl fluoride (PBSF), a lower yield (82%) of 3c was obtained (Table 3, entry 8); however, PBSF was found to give an elimination by-product as mentioned before (Table 2, entry 6). Finally, when SO2F2 was used under similar conditions, a low yield (12%) of azide 3c was formed (Table 3, entry 9). Clearly, SulfoxFluor was superior to other sulfonimidoyl fluorides and sulfonyl fluorides in the present deoxyazidation reaction. It is interesting to note that the use of bis(2,4-dichlorophenyl) chlorophosphate ((2,4-Cl2C6H3O)2P(O)Cl, 1j)/NaN3/DMAP, a state-of-the-art method for deoxyazidation of alcohols at room temperature30, failed to convert 2,2,2-trifluoroethanol (2c) into azide 3c (Table 3, entry 10; for details, see the Supplementary Methods).
Deoxyazidation of alcohols
With the optimized conditions (Table 1, entry 4 for primary alcohols; Table 2, entry 5 for secondary alcohols) in hand, we investigated the substrate scope of this SulfoxFluor-mediated deoxyazidation reaction using NaN3 as an azide source. The results are shown in Fig. 2. Fifty structurally diverse primary and secondary alcohols were applied in this reaction, nearly half of which are pharmaceutically important molecules. In most cases, the corresponding alkyl azides were obtained in good or excellent yields. The reaction tolerates a variety of functional groups, such as aldehydes, alkenes, alkynes, ketones, esters, amides, halides, nitro, and sulfonyl groups (see Fig. 2). It has been known that aldehydes are not amenable to Mitsunobu reactions owing to the condensation of the aldehyde functionality with Huisgen zwitterions48; however, it is remarkable that under our current azidation reaction conditions, desired product 3w was obtained in 82% yield. Our reaction is also compatible with the majority of heterocycles; heteroaromatic substrates such as indole, benzothiazole, pyridine, thiazole, thiophene, benzothiophene, and pyrimidine are all suitable substrates for this reaction (see 3k–3o, 3x, 3y, and 3ae). The reaction of enantiomerically enriched secondary alcohols 2q and 2v proceeded smoothly to give products 3q and 3v in excellent yields (95% and 96%) and stereospecificity (98.6% and >99.9% e.s.) respectively, which is in accordance with an inversion of configuration resulting from an SN2 mechanism (CCDC 2005774 (3v) contains the supplementary crystallographic data for this paper. These data are provided free of charge by The Cambridge Crystallographic Data Centre.)40,42. Similarly, the stereogenic centers of 3ab, 3af, 3ag, 3av, and 3ba were assigned by analogy. Cyclic alcohols such as four-, five-, and six-membered rings could also undergo efficient deoxyazidation under the standard conditions, giving corresponding products in good to excellent yields (3u–3ae). Notably, the carbamate group in primary alcohol 2g is also compatible without elimination and intramolecular cyclization by-products under the present reaction conditions, giving azide 3g in 60% yield.
aIsolated yields. bFor primary alcohols: reactions were conducted on 0.2 mmol scale using 2.2 equiv of SulfoxFluor, 4.0 equiv of NaN3 and 1.8 equiv of DBU. cReaction was conducted on 0.2 mmol scale using 2.5 equiv of SulfoxFluor, 5.0 equiv of NaN3, and 1.8 equiv of DBU. dFor secondary alcohols: reactions were conducted on 0.2 mmol scale using 2.8 equiv of SulfoxFluor, 2.0 equiv of NaN3, and 4.0 equiv of DBU. eThe abbreviation e.s. refers to enantiospecifity, e.s. = (e.e. of 3)/(e.e. of 2) × 100%. fReaction was conducted on a 0.2 mmol scale using 1.05 equiv of 1j, 4.0 equiv of NaN3, and 1.2 equiv of DMAP in DMF (0.2 M) at rt for 12 h. gEpimer ratio >20:1. hReaction was conducted on a 0.4 mmol scale. iReaction was conducted on a 0.16 mmol scale.
Late-stage modification of structurally complex molecules (such as natural products and drugs) can rapidly generate new pharmaceutical candidates with potentially improved pharmacological profiles49,50,51,52,53,54,55,56,57,58. Late-stage azidation is particularly attractive in this regard because the incorporation of azide functionality (followed by click reaction) can quickly build modular molecular libraries. Inspired by these considerations, we applied our deoxyazidation method in the late-stage functionalization of complex molecules such as isosorbide-2-acetate (2af), TF-HF (2ag) and (-)-Corey lactone benzoate (2ah), and the corresponding azidation products 3af, 3ag, and 3ah were obtained in 92%, 92%, and 81% yields, respectively (Fig. 2). The dansyl chloride derivative 2ai was also efficiently deoxyazidated to give product 3ai in 94% yield, indicating that tertiary amine functionality is compatible with the current azidation conditions. It is interesting to mention that, the current deoxyazidation reaction has good selectivity toward multiple alcohols (such as 2ak-2an), that is, the azidation occurs predominantly on the less hindered hydroxyl group and affords the mono-azidation products in good yields (see 3ak-3an). The drug derivatives estrone (2aq), oxaprozin (2ar), adenosine (2as), indometacin (2at), bendazac (2au), DL-α-tocopherol (2aw), estradiol (2ax), and glucose derivative 2av were all able to undergo deoxyazidation smoothly to afford the corresponding products 3aq-3ax in moderate to good yields. Most remarkably, when antifungal drug posaconazole (2ba), the most complex molecule among the fifty substrates shown in Fig. 2, was subjected to the current deoxyazidation process, product 3ba was isolated in satisfactory yield (47%).
Of note that our synthetic method is also advantageous over the previously reported deoxyazidation system (2,4-Cl2C6H3O)2P(O)Cl/NaN3/DMAP in converting normal secondary alcohols to organoazides. For example, starting from alcohol 2r, the use of SulfoxFluor could afford the corresponding azide 3r in nearly quantitative yield, whereas the use of (2,4-Cl2C6H3O)2P(O)Cl under reported standard conditions30 provided 3r in low yield (21%) even prolonging the reaction time to 12 h. In the latter case, both the bis(2,4-dichlorophenyl) phosphate intermediate and unreacted alcohol 2r were isolated (for detail, see the Supplementary Methods), indicating the low efficiency of (2,4-Cl2C6H3O)2P(O)Cl in the activation of normal secondary alcohols.
Synthetic applications
To further demonstrate the synthetic utility of the current deoxyazidation protocol, we carried out the gram-scale synthesis. As shown in Fig. 3a, the deoxyazidation reaction of dansyl chloride derivative 2ai was carried out under standard conditions. This reaction proceeded well and afforded the desired azide 3ai in 94% yield. Remarkably, the azide product 3ai could be converted to triazole 9 under copper catalysis59 in nearly quantitative yield, which significantly increased the complexity of the molecule and demonstrated the potential application of this azidation protocol in drug discovery. Furthermore, indometacin derivative 2at was subjected to the deoxyazidation reaction and subsequent click reaction in one pot, and triazole 10 was obtained in 83% overall yield (Fig. 3b; for details, see the Supplementary Methods).
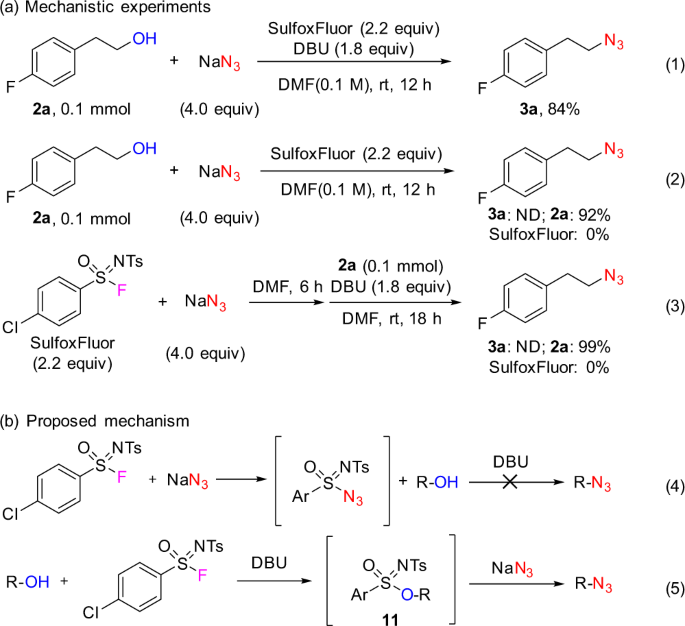
Experimental investigation of a reaction mechanism
Control experiments were performed to investigate the reaction mechanism (Fig. 4). Alcohol 2a reacted under standard conditions to give azide 3a in 84% yield (determined by 19F NMR; Fig. 4, Eq 1). However, when DBU was not added, the expected product 3a was not detected and the alcohol 2a remained, along with the complete consumption of SulfoxFluor (Fig. 4, Eq 2). This result indicates that SulfoxFluor itself could react with NaN3. Further experiments showed the pre-formed sulfonimidoyl azide intermediate was not able to undergo the desired deoxyazidation reaction (Fig. 4, Eqs 3 and 4). Based on the above-mentioned experiments, a plausible reaction mechanism is proposed for the deoxyazidation of alcohols with SulfoxFluor (Fig. 4, Eq 5). The activated alcohol (by DBU) reacts with SulfoxFluor to form sulfonimidate ester 11, which undergoes a nucleophilic displacement of the sulfonimidate group by azide ion to give the corresponding alkyl azide. The success of the azidation reaction lies in better nucleophilicity of azide ion over that of fluoride ion.
a The comparison of the standard experiment and the control experiments. Eq 1 refers to the deoxyazidation reaction conducted under the standard conditions. Eq 2 refers to the control experiment performed in the absence of DBU. Eq 3 refers to the control experiment performed via reverse addition of the reactants. b Proposed mechanism of competitive consumption of SulfoxFluor by NaN3 (Eq 4) and the desired deoxyazidation of alcohols (Eq 5). ND not detected.
Discussion
We have developed a general protocol for the direct deoxyazidation of alcohols with NaN3, which provides a powerful tool to synthesize structurally diverse alkyl azides from readily available alcohols under mild conditions. Our previously developed SulfoxFluor reagent60,61 plays an important role in this efficient deoxyazidation reaction. To our knowledge, the substrate scope and functional group tolerance of this method are superior to those of other deoxyazidation reactions (starting from alcohols) reported to date. Moreover, we have shown that this method can be applied in the late-stage modification of natural products and pharmaceutically relevant molecules, showcasing that this protocol promises to find practical applications in life sciences and related fields. Further exploration in this direction is underway in our laboratory.
Methods
General
The general procedures for deoxyazidation of primary alcohols 2 with SulfoxFluor 1a are as follows. In a typical experiment, into a 25-mL Schlenk tube (glass) were sequentially added 2-(naphthalen-1-yl)ethan-1-ol 2c (34.4 mg, 0.2 mmol), SulfoxFluor (152.9 mg, 0.44 mmol, 2.2 equiv), NaN3 (52.0 mg, 0.8 mmol), DMF (2.0 mL), and DBU (54 μL, 0.36 mmol, 1.8 equiv) under N2 atmosphere. The mixture was stirred at room temperature for 12 h. Then water (2–5 mL) was added and the mixture was extracted with Et2O (3 × 2 mL). The combined organic layers were dried over Na2SO4, filtered, concentrated under reduced pressure, and purified by chromatography on silica gel to afford alkyl azide 3c (37.7 mg, 96%). The deoxyazidation of secondary alcohols 2 with SulfoxFluor 1a were carried out similarly and the procedures are presented in Supplementary Methods.
Data availability
The authors declare that the data supporting the findings of this study are available within the article and its Supplementary Information files. For the experimental procedures, and spectroscopic and physical data of compounds, see Supplementary Methods. For NMR analysis of the compounds in this article, see Supplementary Figs. 1–134. The CCDC 2005774 [https://doi.org/10.5517/ccdc.csd.cc25b5d3] contains the crystallographic data for compound 3v (Supplementary Fig. 139; Supplementary Table 6). These data can be obtained free of charge from the Cambridge Crystallographic Data Center (www.ccdc.cam.ac.uk).
References
Bräse, S. & Banert, K. Organic Azides: Syntheses and Applications (Wiley, 2010).
Patai, S. The Chemistry of the Azide Group (Interscience, 1971).
Bayley, H. & Knowles, J. R. Photoaffinity labeling. Methods Enzymol. 46, 69–114 (1977).
Bayley, H. Photogenerated Reagents in Biochemistry and Molecular Biology (Elsevier, 1983).
Fedan, J. S., Hogaboom, G. K. & O’Donnell, J. P. Photoafflnity labeling as pharmacological tools. Biochem. Pharmacol. 33, 1167–1180 (1984).
Radominska, A. & Rdrake, R. Synthesis and uses of azido-substituted nucleoside diphosphate sugar photoaffinity analogs. Methods Enzymol. 230, 330–339 (1994).
Singh, A., Thornton, E. R. & Westheimer, F. H. Photolysis of diazoacetylchymotrypsin. J. Biol. Chem. 237, 3006–3008 (1962).
Kolb, H. C., Finn, M. & Sharpless, G. K. B. Click chemistry: diverse chemical function from a few good reactions. Angew. Chem. Int. Ed. 40, 2004–2021 (2001).
Bock, V. D., Hiemstra, H. & van Maarseveen, J. H. CuI-Catalyzed alkyne–azide “click” cycloadditions from a mechanistic and synthetic perspective. Eur. J. Org. Chem. 1, 51–68 (2006).
Becer, C. R., Hoogenboom, R. & Schubert, U. S. Click chemistry beyond metal-catalyzed cycloaddition. Angew. Chem. Int. Ed. 48, 4900–4908 (2009).
Meng, G. et al. Modular click chemistry libraries for functional screens using a diazotizing reagent. Nature 574, 86–89 (2019).
Bryan, M. C. et al. Key green chemistry research areas from a pharmaceutical manufacturers’ perspective revisited. Green. Chem. 20, 5082–5013 (2018).
Mitsunobu, O., Wada, M. & Sano, T. Stereospecific and stereoselective reactions. I. Preparation of amines from alcohols. J. Am. Chem. Soc. 94, 679–680 (1972).
Hughes, D. L. The Mitsunobu reaction. Org. React. 42, 335–656 (1992).
Loibner, H. & Zbiral, E. New preparative methods using triphenylphosphine-diethyl-azodicarboxylate-hydroxycompound. Helv. Chim. Acta 60, 417–425 (1977).
Mitsunobu, O. The use of diethyl azodicarboxylate and triphenylphosphine in synthesis and transformation of natural products. Synthesis 1981, 1–28 (1981).
Hughes, D. L. Progress in Mitsunobu reaction: a review. Org. Prep. Proced. Int. 28, 127–164 (1996).
Saito, A., Saito, K., Tanaka, A. & Oritani, T. An efficient method for converting alcohols to azides with 2,4,4,6-tetrabromo-2,5-cyclohexadienone/PPh3/Zn(N3)2·2Py. Tetrahedron Lett. 38, 3955–3958 (1997).
Mizuno, M. & Shior, T. Efficient method for the one-pot azidation of alcohols using bis(p-nitrophenyl) phosphorazidate. Chem. Commun. 22, 2165–2166 (1997).
Fabiano, E., Golding, B. T. & Sadeghi, M. M. A simple conversion of alcohols into amines. Synthesis 1987, 190–192 (1987).
Bessodes, M., Abushanab, E. & Antonakis, K. Enantiospecific synthesis of the immunopotentiators erythro-9 (2-hydroxy-3-nonyl) hypoxanthines and the threo-diastereomers. Tetrahedron Lett. 25, 5899–5902 (1984).
Lee, S.-H., Yoon, J., Chung, S.-H. & Lee, Y.-S. Efficient asymmetric synthesis of 2,3-diamino-3-phenylpropanoicacid derivatives. Tetrahedron 57, 2139–2145 (2001).
He, L., Wanunu, M., Byun, H.-S. & Bittman, R. Regioselective and stereospecific azidation of 1,2- and 1,3-diols by azidotrimethylsilane via a Mitsunobu reaction. J. Org. Chem. 64, 6049 (1999).
Lal, B., Pramanik, B. N., Manhas, M. S. & Bose, A. K. Diphenylphosphoryl azide a novel reagent for the stereospecific synthesis of azides from alcohols. Tetrahedron Lett. 18, 1977–1980 (1977).
Viaud, M. C. & Rollin, P. Zinc azide mediated Mitsunobu substitution. An expedient method for the one-pot azidation of alcohols Synthesis 1990, 130-132 (1990).
Iranpoor, N., Firouzabadi, H., Akhlaghinia, B. & Nowrouzi, N. A novel and highly selective conversion of alcohols, thiols, and silyl ethers to azides using the triphenylphosphine/2,3-dichloro-5,6-dicyanobenzoquinone(DDQ)/n-Bu4NN3 system. Tetrahedron Lett. 45, 3291–3294 (2004).
Kumar, H. M. S., Reddy, B. V. S., Anjaneyulu, S. & Yadav, J. S. An expedient and highly selective conversion of alcohols to azides using a NaN3/BF3•Et2O System. Tetrahedron Lett. 39, 7385–7388 (1998).
Jayanthi, A., Gumaste, V. K. & Deshmukh, A. R. A. S. A simple one-pot method for the preparation of allyl azides from allyl alcohols using triphosgene: synthesis of N1-cinnamyl azetidin-2-ones. Synlett 2004, 979–982 (2004).
Rad, M. N. S., Behrouz, S. & Khalafi-Nezhad, A. A simple one-pot procedure for the direct conversion of alcohols into azides using TsIm. Tetrahedron Lett. 48, 3445- (2007).
Yu, C., Liu, B. & Hu, L. A simple one-pot procedure for the direct conversion of alcohols to azides via phosphate activation. Org. Lett. 2, 1959–1961 (2000).
Li, Z., Qiao, R., Yang, Z., Zhang, L. & Zhang, L. One-step synthesis of novel tricyclic isomeric azidonucleosides. Tetrahedron. Asymmetry 17, 1056–1061 (2006).
Toyota, M., Komori, C. & Ihara, M. A concise formal total synthesis of mappicine and nothapodytine B via an intramolecular hetero Diels−Alder reaction. J. Org. Chem. 65, 7110–7113 (2000).
Reddy, G. V. S., Rao, G. V., Subramanyam, R. V. K. & Iyengar, D. S. A new novel and practical one pot methodology for conversion of alcohols to amines. Synth. Commun. 30, 2233–2237 (2000).
Chen, J., Lin, J. & Xiao, J. Dehydroxylation of alcohols for nucleophilic substitution. Chem. Commun. 54, 7034–7037 (2018).
Reggelin, M. & Zur, C. Sulfoximines: structures, properties and synthetic applications. Synthesis 2000, 1–64 (2000).
Lücking, U. Sulfoximines: a neglected opportunity in medicinal chemistry. Angew. Chem. Int. Ed. 52, 9399–9408 (2013).
Bizet, V., Kowalczyk, R. & Bolm, C. Fluorinated sulfoximines: syntheses, properties and applications. Chem. Soc. Rev. 43, 2426–2438 (2014).
Shen, X. & Hu, J. Fluorinated sulfoximines: preparation, reactions and applications. Eur. J. Org. Chem. 2014, 4437–4451 (2014).
Liu, Q., Shen, X., Ni, C. & Hu, J. Stereoselective carbonyl olefination with fluorosulfoximines: facile access to Z or E terminal monofluoroalkenes. Angew. Chem. Int. Ed. 56, 619–623 (2017).
Guo, J. et al. Rapid deoxyfluorination of alcohols with N-Tosyl-4-chlorobenzenesulfonimidoyl fluoride (SulfoxFluor) at room temperature. Chem. Eur. J. 25, 7259–7264 (2019).
Tang, H., Cheng, J., Liang, Y. & Wang, Y. Discovery of a chiral fluorinated azetidin-2-one as a tubulin polymerisation inhibitor with potent antitumour efficacy. Eur. J. Med. Chem. 197, 112323 (2020).
Ouellette, R. J. & Rawn, J. D. Organic Chemistry: Structure, Mechanism, and Synthesis 2nd edn (Elsevier, 2018).
Nielsen, M. K., Ugaz, C. R., Li, W. & Doyle, A. G. PyFluor: a low-cost, stable, and selective deoxyfluorination reagent. J. Am. Chem. Soc. 137, 9571–9574 (2015).
Nielsen, M. K., Ahneman, D. T., Riera, O. & Doyle, A. G. Deoxyfluorination with sulfonyl fluorides: navigating reaction space with machine learning. J. Am. Chem. Soc. 140, 5004–5008 (2018).
Bennua-Skalmowski, B. & Vorbrüggen, H. A facile conversion of primary or secondary alcohols with n-perfluorobutane-sulfonyl fluoride/1,8-diazabicyclo[5.4.0]undec-7-ene into their corresponding fluorides. Tetrahedron Lett. 36, 2611–2614 (1995).
Vorbrüggen, H. The conversion of primary or secondary alcohols with nonaflyl fluoride into their corresponding inverted fluorides. Synthesis 2008, 1165–1174 (2008).
Yin, J., Zarkowsky, D. S., Thomas, D. W., Zhao, M. M. & Huffman, M. A. Direct and convenient conversion of alcohols to fluorides. Org. Lett. 6, 1465–1468 (2004).
Girard, M., Murphy, P. & Tsou, N. N. An exception to the normal Mitsunobu reaction with phenols; the formation of hydrazones from salicylaldehydes. Tetrahedron Lett. 46, 2449 (2005).
Long, A. A. W., Nayler, J. H. C., Smith, H., Taylor, T. & Ward, N. Derivatives of 6-aminopenicillanic acid. Part X1. a-amino-p-hydroxy-benzylpenicillin. J. Chem. Soc. C 1920–1922 (1971).
Lewis, C. A. & Miller, S. J. Site-selective derivatization and remodeling of erythromycin A by using simple peptide-based chiral catalysts. Angew. Chem. Int. Ed. 45, 5616–5619 (2006).
Newman, D. J. & Cragg, G. M. Natural products as sources of new drugs over the last 25 years. J. Nat. Prod. 70, 461–477 (2007).
Butler, M. S. Natural products to drugs: natural product-derived compounds in clinical trials. Nat. Prod. Rep. 25, 475–516 (2008).
Reed, S. A., Mazzotti, A. R. & White, M. C. A catalytic, Brønsted base strategy for intermolecular allylic C−H amination. J. Am. Chem. Soc. 131, 11701–11706 (2009).
Balthaser, B. R., Maloney, M. C., Beeler, A. B., Porco, J. A. Jr. & Snyder, J. K. Remodelling of the natural product fumagillol employing a reaction discovery approach. Nat. Chem. 3, 969–973 (2011).
Jordan, P. A. & Miller, S. J. An approach to the site-selective deoxygenation of hydroxy groups based on catalytic phosphoramidite transfer. Angew. Chem. Int. Ed. 51, 2907–2911 (2012).
Pathak, T. P. & Miller, S. J. Site-selective bromination of vancomycin. J. Am. Chem. Soc. 134, 6120–6123 (2012).
Fowler, B. S., Laemmerhold, K. M. & Miller, S. J. Catalytic site-selective thiocarbonylations and deoxygenations of vancomycin reveal hydroxyl-dependent conformational effects. J. Am. Chem. Soc. 134, 9755–9761 (2012).
Voica, A.-F., Mendoza, A., Gutekunst, W. R., Fraga, J. O. & Baran, P. S. Guided desaturation of unactivated aliphatics. Nat. Chem. 4, 629–635 (2012).
Rostovtsev, V. V., Green, L. G., Fokin, V. V. & Sharpless, K. B. A stepwise Huisgen cycloaddition process: copper(I)-catalyzed regioselective “ligation” of azides and terminal alkynes. Angew. Chem. Int. Ed. 41, 2596–2599 (2002).
Liu, R. et al. Modified and scalable synthesis of N-tosyl-4-chlorobenzenesulfonimidoyl fluoride (SulfoxFluor): direct imidation of sulfinyl chlorides with chloramine-T trihydrate. Org. Process Res. Dev. 26, 380–386 (2022).
Zhou, X. et al. Method for preparing a deoxyfluorination reagent. Chinese patent application. CN 113717087A (2020).
Acknowledgements
Financial support for this work by the National Key Research and Development Program of China (2021YFF0701700 and 2016YFB0101200), the National Natural Science Foundation of China (21632009), the Key Programs of the Chinese Academy of Sciences (KGZD-EW-T08), the Key Research Program of Frontier Sciences of CAS (QYZDJ-SSW-SLH049), and Shanghai Science and Technology Program (18JC1410601) is acknowledged. J.G. thanks Jie Sun (SIOC) for assistance with the X-ray crystallographic analysis.
Author information
Authors and Affiliations
Contributions
J.H. conceived the project. J.H., J.G., X.W., and C.N. designed the experiments, analyzed the data, and co-wrote the manuscript. J.G. and X.W. performed synthetic and mechanistic experiments. X.(L.)W. tested the e.e. value. J.H., J.G., X.W., and C.N. discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks the anonymous reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Guo, J., Wang, X., Ni, C. et al. SulfoxFluor-enabled deoxyazidation of alcohols with NaN3. Nat Commun 13, 2752 (2022). https://doi.org/10.1038/s41467-022-30132-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-022-30132-x
This article is cited by
-
Multipurpose sulfoximine-mediated radical γ-heteroarylation of unactivated C(sp3)-H bonds
Science China Chemistry (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.